Avem de învațat atâta timp cât trăim, poate doar nu știm unde să privim, sau ...
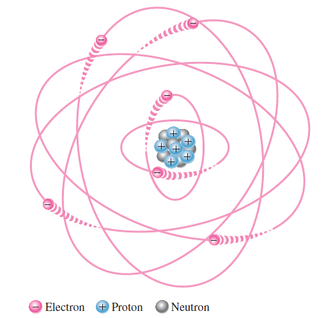
The Bohr model
Atoms are extremely small and cannot be seen even with the strongest optical microscopes; however, a scanning tunneling microscope can detect a single atom. The nucleus is so small and the electrons orbit at such distances that the atom is mostly empty space. To put it in
perspective, if the proton in a hydrogen atom were the size of a golf ball, the electron orbit would be approximately one mile away.
Protons and neutrons are approximately the same mass. The mass of an electron is 1 1836 of a proton. Within protons and neutrons there are even smaller particles called quarks.
Atomii sunt extrem de mici și nu pot fi văzuți chiar și cu cele mai puternice microscoape optice; totuși, un microscop de scanare poate detecta un singur atom. Nucleul este atât de mic, iar electronii orbitează la astfel de distanțe încât atomul este în mare parte spațiu gol. Pentru a pune în perspectivă, dacă protonul dintr-un atom de hidrogen avea dimensiunea unei mingi de golf, orbita de electroni ar fi la aproximativ o milă distanță.
Protonii și neutronii sunt aproximativ de aceeași masă. Masa unui electron este de 1/1836 de proton. În protoni și neutroni există chiar și particule mai mici numite quarci.
Atoms are extremely small and cannot be seen even with the strongest optical microscopes; however, a scanning tunneling microscope can detect a single atom. The nucleus is so small and the electrons orbit at such distances that the atom is mostly empty space. To put it in
perspective, if the proton in a hydrogen atom were the size of a golf ball, the electron orbit would be approximately one mile away.
Protons and neutrons are approximately the same mass. The mass of an electron is 1 1836 of a proton. Within protons and neutrons there are even smaller particles called quarks.
Atomii sunt extrem de mici și nu pot fi văzuți chiar și cu cele mai puternice microscoape optice; totuși, un microscop de scanare poate detecta un singur atom. Nucleul este atât de mic, iar electronii orbitează la astfel de distanțe încât atomul este în mare parte spațiu gol. Pentru a pune în perspectivă, dacă protonul dintr-un atom de hidrogen avea dimensiunea unei mingi de golf, orbita de electroni ar fi la aproximativ o milă distanță.
Protonii și neutronii sunt aproximativ de aceeași masă. Masa unui electron este de 1/1836 de proton. În protoni și neutroni există chiar și particule mai mici numite quarci.
The Quantum Model
Although the Bohr model of an atom is widely used because of its simplicity and ease of visualization, it is not a complete model. The quantum model, a more recent model, is considered to be more accurate. The quantum model is a statistical model and very difficult to understand or visualize. Like the Bohr model, the quantum model has a nucleus of protons and neutrons surrounded by electrons. Unlike the Bohr model, the electrons in the quantum model do not exist in precise circular orbits as particles. Two important theories underlie
the quantum model: the wave-particle duality and the uncertainty principle.
Atomic orbitals do not resemble a discrete circular path for the electron as depicted in Bohr’s planetary model. In the quantum picture, each shell in the Bohr model is a threedimensional space surrounding the atom that represents the mean (average) energy of the electron cloud. The term electron cloud (probability cloud) is used to describe the area around an atom’s nucleus where an electron will probably be found.
Although the Bohr model of an atom is widely used because of its simplicity and ease of visualization, it is not a complete model. The quantum model, a more recent model, is considered to be more accurate. The quantum model is a statistical model and very difficult to understand or visualize. Like the Bohr model, the quantum model has a nucleus of protons and neutrons surrounded by electrons. Unlike the Bohr model, the electrons in the quantum model do not exist in precise circular orbits as particles. Two important theories underlie
the quantum model: the wave-particle duality and the uncertainty principle.
Atomic orbitals do not resemble a discrete circular path for the electron as depicted in Bohr’s planetary model. In the quantum picture, each shell in the Bohr model is a threedimensional space surrounding the atom that represents the mean (average) energy of the electron cloud. The term electron cloud (probability cloud) is used to describe the area around an atom’s nucleus where an electron will probably be found.
Does an Electron 'particle' exist?
Tesla, Heaviside, JC Maxwell & CP Steinmetz said NO
Nikola Tesla November 1928 interview: On the whole subject of matter, in fact, Dr. Tesla holds views that are startlingly original. He disagrees with the accepted atomic theory of matter, and does not believe in the existence of an “electron” as pictured by science.
“To account for its apparently small mass, science conceives of the electron as a hollow sphere, a sort of bubble, such a bubble could exist in a medium as a gas or liquid because its internal pressure is not altered by deformation. But if, as supposed, the internal pressure of an electron is due to the repulsion of electric masses, the slightest conceivable deformation must result in the destruction of the bubble! Just to mention another improbability...” - Nikola Tesla Article: “A Famous Prophet of Science Looks into the Future” (Popular Science Monthly)
“My ideas regarding the electron are at variance with those generally entertained. I hold that it is a relatively large entity carrying a surface charge and is not an elementary unit (particle). When the ‘electron’ leaves an electrode of high potential and in a high vacuum it carries an electrostatic charge many times greater than normal.” – N. Tesla
“In the theoretical treatment of these electrons we are faced with the difficulty that electro-dynamic theory by itself is unable to give an account of their nature.” “For since electrical masses constituting the electron would necessarily be scattered under the influence of their mutual repulsions, unless there are forces of another kind operating between them the nature of which has hitherto remained obscure to us.” - Einstein on electrons; “Relativity”, by Albert Einstein, Random House Publisher, 1916
“To describe an electron as a negatively charged body is equivalent to saying that it is an expanding-contracting particle. There is no such condition in nature as a negative charge, nor are there negatively charged particles. Charge and discharge are opposite conditions, as filling and emptying, or compressing and expanding are opposite conditions.” – W. Russell
JJ Thomson developed the “Ether Atom” ideas of M. Faraday into his “Electronic Corpuscle”, this indivisible unit. One corpuscle terminates on one Faradic tube of force, and this quantifies as one Coulomb. This corpuscle is not and electron, it is a constituent of what today is known incorrectly as an “electron”. (Thomson relates 1000 corpuscles per electron) In this view, that taken by W. Crookes, J.J. Thomson, and N. Tesla, the cathode ray is not electrons, but in actuality corpuscles of the Ether.” – E. Dollard
“There is no rest mass to an ‘electron’. It is given here the ‘electron’ is no more than a broken loose “hold fast” under the grip of the tensions within the dielectric lines of force. They are the broken ends of the split in half package of spaghetti. Obviously this reasoning is not welcome in the realm of Einstein’s Theory of Relativity.” – E. Dollard
“Unfortunately to a large extent in dealing with dielectric fields the prehistoric conception of the electro-static charge, the ‘electron’, on the conductor still exists, and by its use destroys the analogy between the two components of the electric field, the magnetic and dielectric. This makes the consideration of dielectric fields unnecessarily complicated” - C.P. Steinmetz (Electric Discharges, Waves and Impulses)
The idea of electricity as a flow of ‘electrons’ in a conductor was regarded by Oliver Heaviside as “a psychosis”. This encouraged Heaviside to begin a series of writings
Also consider the J.J. Thomson concept of the "electron" (his own discovery). Thomson considered the electron the terminal end of one unit line of dielectric induction.
“Electrons as a separate, distinct entity…doesn’t really exist, they are merely bumps in something called a ‘field’.” - Dr. Steve Biller
Tesla, Heaviside, JC Maxwell & CP Steinmetz said NO
Nikola Tesla November 1928 interview: On the whole subject of matter, in fact, Dr. Tesla holds views that are startlingly original. He disagrees with the accepted atomic theory of matter, and does not believe in the existence of an “electron” as pictured by science.
“To account for its apparently small mass, science conceives of the electron as a hollow sphere, a sort of bubble, such a bubble could exist in a medium as a gas or liquid because its internal pressure is not altered by deformation. But if, as supposed, the internal pressure of an electron is due to the repulsion of electric masses, the slightest conceivable deformation must result in the destruction of the bubble! Just to mention another improbability...” - Nikola Tesla Article: “A Famous Prophet of Science Looks into the Future” (Popular Science Monthly)
“My ideas regarding the electron are at variance with those generally entertained. I hold that it is a relatively large entity carrying a surface charge and is not an elementary unit (particle). When the ‘electron’ leaves an electrode of high potential and in a high vacuum it carries an electrostatic charge many times greater than normal.” – N. Tesla
“In the theoretical treatment of these electrons we are faced with the difficulty that electro-dynamic theory by itself is unable to give an account of their nature.” “For since electrical masses constituting the electron would necessarily be scattered under the influence of their mutual repulsions, unless there are forces of another kind operating between them the nature of which has hitherto remained obscure to us.” - Einstein on electrons; “Relativity”, by Albert Einstein, Random House Publisher, 1916
“To describe an electron as a negatively charged body is equivalent to saying that it is an expanding-contracting particle. There is no such condition in nature as a negative charge, nor are there negatively charged particles. Charge and discharge are opposite conditions, as filling and emptying, or compressing and expanding are opposite conditions.” – W. Russell
JJ Thomson developed the “Ether Atom” ideas of M. Faraday into his “Electronic Corpuscle”, this indivisible unit. One corpuscle terminates on one Faradic tube of force, and this quantifies as one Coulomb. This corpuscle is not and electron, it is a constituent of what today is known incorrectly as an “electron”. (Thomson relates 1000 corpuscles per electron) In this view, that taken by W. Crookes, J.J. Thomson, and N. Tesla, the cathode ray is not electrons, but in actuality corpuscles of the Ether.” – E. Dollard
“There is no rest mass to an ‘electron’. It is given here the ‘electron’ is no more than a broken loose “hold fast” under the grip of the tensions within the dielectric lines of force. They are the broken ends of the split in half package of spaghetti. Obviously this reasoning is not welcome in the realm of Einstein’s Theory of Relativity.” – E. Dollard
“Unfortunately to a large extent in dealing with dielectric fields the prehistoric conception of the electro-static charge, the ‘electron’, on the conductor still exists, and by its use destroys the analogy between the two components of the electric field, the magnetic and dielectric. This makes the consideration of dielectric fields unnecessarily complicated” - C.P. Steinmetz (Electric Discharges, Waves and Impulses)
The idea of electricity as a flow of ‘electrons’ in a conductor was regarded by Oliver Heaviside as “a psychosis”. This encouraged Heaviside to begin a series of writings
Also consider the J.J. Thomson concept of the "electron" (his own discovery). Thomson considered the electron the terminal end of one unit line of dielectric induction.
“Electrons as a separate, distinct entity…doesn’t really exist, they are merely bumps in something called a ‘field’.” - Dr. Steve Biller